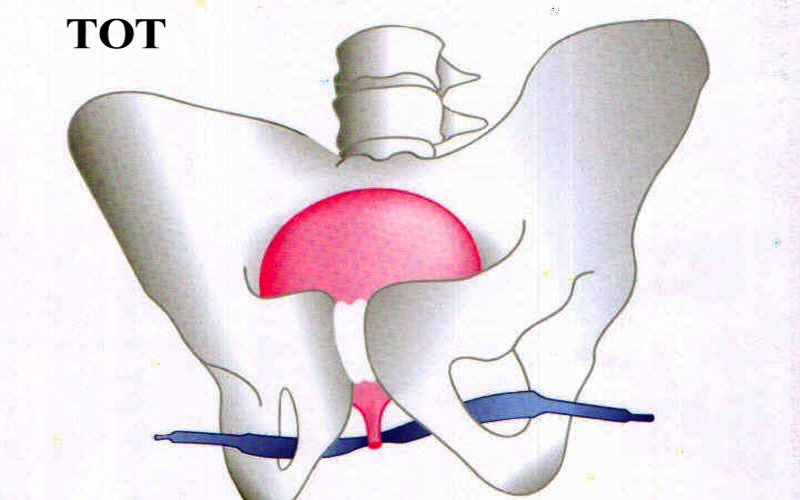
Transvaginal tape is a polypropylene (PP) woven fabric used to shore up dropped pelvic organs known as pelvic organ prolapse (POP), or stress urinary incontinence (SUI), the involuntary leakage of urine.
The tape is formed into a ribbon, at least 12 inches in length, and placed under the urethra at the midway point to support it, hence the name mid-urethral sling (MUS).
Transvaginal tape, also called tension-free tape (TVT), supports the urethra and exits in the abdomen near the belly button while the transvaginal tape obturator (TVT-O) is similar in its function except it exits through the groin area after passing through the obturator foramen.
Both meshes are intended to be a permanent implant, despite what a woman maybe told by her doctor.
A population-based study, presented at the 2015 International Continence Society in Montreal, and published in the American Journal of Obstetrics and Gynecology in 2016 (Foss Hansen M.), suggests the TOT sling has a greater rate of re-operation than other mid-urethral sling surgeries.
Investigators in Denmark found the transvaginal tape obturator (TVT-O) was linked to a two-fold increased risk of re-operation when compared to the TVT. In general, the rate of re-operation for SUI was 10.2% within five years.
A total of 8,671 women enrolled in the Danish National Patient Registry were part of the study. All had undergone surgery to treat incontinence between 1998 through 2007, and had no previous incontinence surgeries. Among the initial group, 888 women were re-operated on within five years.
Factors such as body mass, severity of symptoms and skill of the surgeon were not analyzed. The study does not specify why a re-operation was needed, whether it was for pain, mesh removal, or to treat incontinence.
Other types of surgery included in this study, besides TVT or TVT-O, were urethral injection therapy and Burch colposuspension.
The median time for the re-operations was one-year for sling surgery, two years for a Burch colposuspension and six months for urethral injection surgery. Most re-operations occurred before the two-year mark after the initial implant.
TOT mesh was introduced in Denmark in 2003 and in the U.S., two years earlier.
This study is notable because it is one of the few on TOT surgery that is considered long-term. Most mesh studies are 12-months or less.
A study previously published in the Cochrane Database Syst Review, October 2009 and July 2015, found no significant difference between a TVT or TOT mesh in terms of re-operation rates. This was a smaller patient base of 7,476 women and lasted for one-year post implant.
The Danish researcher, Dr. Foss Hansen suggests more data should be derived from randomized, controlled trials.
TVT-O has been shown to be defectively designed in at least two product liability trials in the U.S. Still, it remains on the market.
Since TOT mesh was marketed by manufacturers such as Boston Scientific, Johnson & Johnson, and C.R. Bard, we are seeing the fallout from a failure to do such long-term studies.
Instead, real live women have become the test subjects, to their detriment. ###